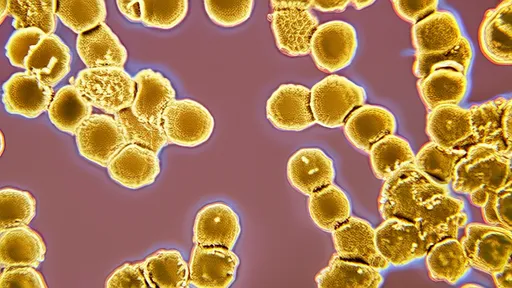
In the silent battleground of petri dishes, an invisible war rages—one that could determine the future of modern medicine. Scientists are meticulously tracking the evolution of antibiotic resistance in bacteria, observing in real-time how these microscopic adversaries adapt to survive our most potent drugs. What begins as a routine experiment often unfolds like a high-stakes thriller, with colonies of bacteria mutating at alarming rates to overcome increasingly concentrated doses of antibiotics.
The process is deceptively simple: a gradient of antibiotic concentration is established across an agar plate, creating zones where only the hardiest microbes can survive. Researchers call this the "microbial evolution experiment," but its implications are anything but academic. Within weeks, bacterial strains that once succumbed to low antibiotic doses develop terrifying resilience, marching across the plate into regions that would have been lethal to their ancestors. This visible manifestation of Darwinian selection reveals why antibiotic resistance has become one of humanity's most pressing public health crises.
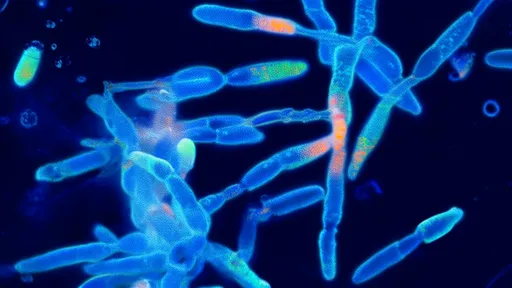
Time-lapse photography captures the drama of bacterial adaptation with startling clarity. What appears as mere discoloration to the naked eye transforms under magnification into advancing fronts of microbial conquest. Some strains develop thicker cell walls; others pump out antibiotics faster than the drugs can accumulate. The most alarming specimens evolve efflux pumps—molecular machines that literally spit antibiotics back out of bacterial cells. These adaptations don't emerge gradually, but in sudden leaps that correspond to genetic mutations visible in genome sequencing.
The evolutionary arms race plays out differently with each antibiotic class. Tetracycline resistance often develops through ribosomal protection—bacteria essentially armor their protein-making machinery. Against fluoroquinolones, microbes frequently mutate their DNA gyrase enzymes, changing the drugs' target sites. Perhaps most disturbingly, some pathogens acquire resistance not through mutation but by horizontal gene transfer—swapping resistance genes like trading cards through plasmids. This bacterial "social network" accelerates resistance spread in ways that terrify epidemiologists.
Real-world parallels emerge from these controlled experiments. The stepwise resistance patterns observed in petri dishes mirror clinical cases where patients' infections become progressively harder to treat. Methicillin-resistant Staphylococcus aureus (MRSA), once confined to hospitals, now circulates in communities—a trajectory predictable from laboratory evolution studies. Carbapenem-resistant Enterobacteriaceae (CRE), dubbed "nightmare bacteria" by the CDC, follow similar adaptive paths in both hospital wards and research incubators.
Beyond documenting the problem, these experiments offer glimmers of hope. By understanding resistance mechanisms, scientists design smarter antibiotic cocktails that attack multiple bacterial vulnerabilities simultaneously. Some teams test "evolution-proof" drugs that force bacteria into evolutionary traps—mutations that confer resistance also cripple virulence. Others explore adjuvant compounds that disable resistance mechanisms, restoring potency to existing antibiotics. The petri dish becomes both crystal ball and testing ground for future therapies.
The most profound revelation isn't about bacteria at all, but about human behavior. Each time researchers observe microbes overcoming another drug, they're witnessing the consequences of antibiotic overuse in medicine and agriculture. These experiments provide visual, visceral proof that every unnecessary prescription and growth-promoting livestock dose accelerates resistance evolution. The bacteria in these plates aren't just surviving—they're holding up a mirror to our collective shortsightedness.
As the experiments continue, new questions emerge about coevolutionary dynamics. How do microbial communities influence individual species' resistance trajectories? Can predatory bacteria or phages alter resistance development? The answers may lie in more complex experimental setups that better mimic human bodies and natural environments. One thing remains clear from these miniature evolutionary theaters: unless we change our approach, the microbes will continue adapting faster than we can develop new drugs.
The petri dish doesn't lie. Its concentric rings of bacterial growth trace the history of our dwindling antibiotic efficacy like tree rings recording climate change. Each experiment ends the same way—with bacteria thriving in antibiotic concentrations that should kill them, a silent warning written in microbial script. For researchers, these plates aren't just laboratory tools but crystal balls showing a future where routine infections become deadly again, unless humanity rises to the evolutionary challenge.

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025
By /Aug 7, 2025

By /Aug 7, 2025
By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025
By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025
By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025